1 Global Medical Device Packaging Market Insight Analysis
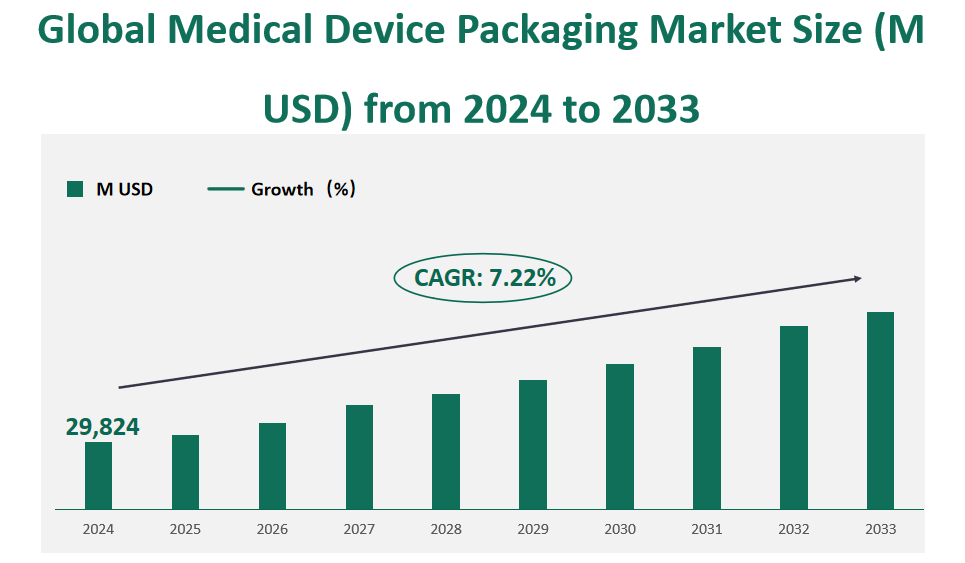
The global medical device packaging market is valued at USD 29,824 million in 2024, with a CAGR of 7.22% from 2024 to 2033.
Medical device packaging refers to the specialized packaging solutions used to protect medical devices from contamination and damage. These packaging solutions are designed to maintain the sterility and integrity of medical devices, ensuring they are safe for use in healthcare settings. The packaging types include trays, pouches, clamshells, and other custom solutions, each tailored to meet specific requirements for different medical devices. The primary applications are sterile and non-sterile packaging, with sterile packaging being a critical segment due to its role in preventing infections and ensuring patient safety.
Figure Global Medical Device Packaging Market Size (M USD) and CAGR (2024-2033)
2 Medical Device Packaging Market Growth Drivers and Restraints
Driving factors: First, high investment in medical technology has driven market growth. With the public’s increased demand for medical care, increased personal income and advances in medical technology, healthcare spending in Europe and other places has continued to rise. This has led to an increase in the number and variety of advanced medical devices on the market, which in turn has driven the demand for medical device packaging. Second, the widespread application of IoT technology in equipment manufacturing and packaging has become an important driving force. In the era of Industry 4.0, IoT technology enables manufacturers to use device data to add value to products.
To ensure the efficiency of the medical device supply chain, smart packaging solutions have been widely adopted, driving market growth. Third, consumers’ attention to product shelf life and ease of use has also promoted market development. To meet demand, companies are actively developing new packaging solutions, such as flexible packaging with excellent barrier properties, which can effectively protect medical devices from moisture and oxidation, and extend shelf life. At the same time, the industry’s application of active, intelligent and antibacterial packaging systems is also increasing, improving the durability of products.
Limiting factors: On the one hand, strict regulatory standards and complex operational issues limit market growth. Globally, governments have set strict regulations and standards for medical device packaging, covering leak detection, transport container performance testing and other aspects. Poorly designed and validated packaging that fails to meet standards not only affects product launches but also damages corporate reputation. In addition, some packaging requires special handling during transportation, which increases costs and operational difficulties.
On the other hand, crude oil price fluctuations and the instability of the petrochemical industry have a negative impact on the market. Crude oil price changes and related geopolitical conflicts have led to unstable raw material supply and prices, increased production costs, affected the supply and demand relationship in the market, and brought challenges to the development of the medical device packaging industry.
3 Technological Innovations in the Medical Device Packaging Market
Technological innovation: In recent years, technological innovation in the medical device packaging market has mainly focused on regulatory standard adjustments and the research and development of new anti-counterfeiting packaging materials. Many countries and regions have adjusted medical device management regulations and put forward higher requirements for packaging performance such as compression resistance, impact resistance, and sterile barrier. Some special medical devices also require outer packaging with anti-oxygen penetration and waterproof functions. This has prompted companies to increase R&D investment and improve the quality and safety of packaging.
At the same time, the research and development of new anti-counterfeiting packaging materials has become a hot topic. Unique outer packaging helps users identify products and reduce the market share of counterfeit and shoddy products. Companies improve the anti-counterfeiting performance of packaging and protect consumer rights and corporate interests by adopting new materials and technologies, such as special printing processes and smart labels.
Corporate mergers and acquisitions: Corporate mergers and acquisitions are also active in the medical device packaging market. For example, after Amcor acquired Bemis, it was able to introduce Bemis’ key medical packaging solutions and technologies into the European market, providing local customers with a wider product portfolio and global support. Amcor has improved its competitiveness in medical sterile barrier applications by using Bemis’ high-performance Tyvek adhesive and CR27 heat-sealing coating technology.
Spectrum Plastics Group acquires PeelMaster Medical Packaging, strengthening its capabilities in the field of sterile medical flexible packaging. PeelMaster has extensive experience in the field of sterile flexible packaging and can produce a variety of medical packaging products. This acquisition will help Spectrum Plastics Group expand its business scope and increase its market share. These mergers and acquisitions and restructuring activities promote resource integration, enable enterprises to acquire new technologies, expand market coverage, enhance competitiveness in the global market, and promote the gradual increase of industry concentration.
4 Global Medical Device Packaging Market Size by Type
Trays are a crucial component of medical device packaging, particularly for sterile products. They are designed to hold, protect, and ensure the safe transportation of medical devices. In 2024, the revenue generated from trays is projected to be $10,978 million USD. This type of packaging is preferred for its ability to maintain sterility and provide a secure, organized solution for medical devices. Trays are commonly used for high-risk medical products such as artificial hips and heart valves, ensuring they remain uncontaminated until use.
Pouches are another significant segment within the medical device packaging market. They are versatile and widely used for various medical devices, ranging from small instruments to larger equipment. In 2024, the revenue from pouches is expected to reach $11,641 million USD. Pouches are known for their ability to protect against contamination and maintain the integrity of medical devices. They are particularly useful for applications requiring sterilization, as they can be designed to withstand various sterilization methods while keeping the contents sterile.
Clamshells are rigid, durable packaging solutions that provide excellent protection and security for medical devices. They are designed to be easy to open and ensure that the devices inside remain undamaged. In 2024, the revenue from clamshells is projected to be $4,481 million USD. Clamshells are often used for products that require a high level of protection during transportation and storage. Their rigid structure and secure closure mechanisms make them ideal for a wide range of medical devices, from small components to larger instruments.
Table Global Medical Device Packaging Market Size by Type in 2024
Type | Market Size (M USD) 2024 |
|---|---|
Trays | 10978 |
Pouches | 11641 |
Clamshell | 4481 |
Others | 2723 |
5 Global Medical Device Packaging Market Size by Application
In 2024, the revenue of the sterile packaging segment is expected to be 25842 million USD, while the non – sterile packaging segment is projected to reach 3982 million USD.
Sterile packaging plays a vital role in the medical device industry. It is designed to prevent the spread of microorganisms and protect medical devices from external contamination. This type of packaging is essential for ensuring the safety and efficacy of medical products, especially those used in invasive procedures. The high demand for sterile packaging is driven by the increasing prevalence of surgeries and the growing need for strict infection control measures in healthcare facilities. For example, in North America, the revenue from sterile packaging in 2024 is expected to be a significant portion of the total medical device packaging revenue, given the region’s advanced healthcare infrastructure and high – volume medical procedures.
On the other hand, non – sterile packaging is also an important part of the market. Although its revenue is lower compared to sterile packaging, it still has a crucial role. Non – sterile packaging is mainly used for devices that do not require strict sterility during storage but need to maintain a certain level of cleanliness. It must meet specific requirements to minimize the risk of microbial contamination if the device is to be sterilized before use. In 2024, regions with developing healthcare systems, such as parts of Asia – Pacific and South America, may witness growth in the non – sterile packaging market as their medical device manufacturing industries expand.
Table Global Medical Device Packaging Market Size by Application in 2024
Application | Market Size (M USD) 2024 |
|---|---|
Sterile Packaging | 25842 |
Non-sterile Packaging | 3982 |
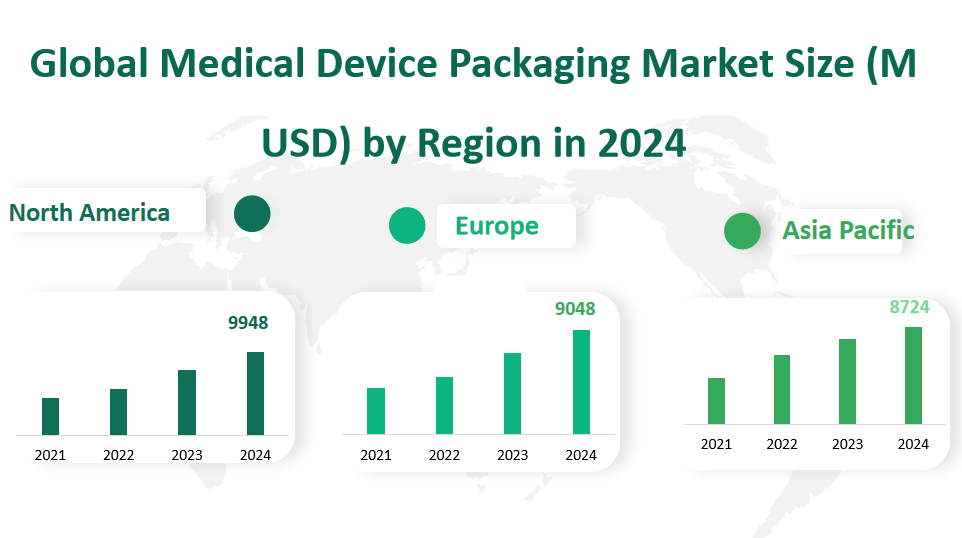
6 Global Medical Device Packaging Market Size by Region
In 2024, the global medical device packaging market shows significant regional variations. North America is expected to have a revenue of 9948 million USD. The region has a well – developed healthcare industry, with a large number of medical device manufacturers and a high demand for advanced packaging solutions. The United States, in particular, is a major contributor to the North American market, accounting for a significant share of the revenue. Its advanced medical technology, high healthcare expenditure, and strict regulatory environment drive the demand for high – quality medical device packaging.
Europe is projected to generate a revenue of 9048 million USD. The European market benefits from its strong manufacturing base, especially in countries like Germany, the UK, and France. These countries have a long – standing reputation for producing high – quality medical devices, and thus, there is a corresponding high demand for reliable packaging solutions. The recent regulatory adjustments in the EU regarding medical device packaging have also spurred innovation and investment in the market.
The Asia – Pacific region is expected to reach a revenue of 8724 million USD in 2024. This region is experiencing rapid economic growth, increasing healthcare spending, and a growing medical device manufacturing industry. China, in particular, is a key market in Asia – Pacific. With its large population and continuous investment in healthcare infrastructure, the demand for medical device packaging in China is rising steadily. Additionally, countries like Japan and South Korea are also significant players, contributing to the region’s growth with their advanced technology and high – quality products.
South America is forecasted to have a revenue of 1231 million USD. Although the market size is relatively smaller compared to the aforementioned regions, it is showing growth potential. Brazil is the largest market in South America, with its expanding healthcare industry and increasing investment in medical research and development driving the demand for medical device packaging.
The Middle East & Africa region is expected to generate a revenue of 872 million USD. Countries like Saudi Arabia, Turkey, and Egypt are leading the growth in this region. The increasing healthcare expenditure and the development of local medical device manufacturing industries in these countries are fueling the demand for medical device packaging. However, the region also faces challenges such as high regulatory standards and supply – chain issues, which may impact the market’s growth to some extent.
Figure Global Medical Device Packaging Market Size (M USD) by Region in 2024
7 Global Medical Device Packaging Market Analysis by Major Players
DowDuPont
Company Profile
DowDuPont is a leading chemical holding company with a rich history dating back to 1897. It specializes in agriculture, materials science, and specialty products, serving a diverse range of industries including healthcare, automotive, and electronics. The company is headquartered in the United States and operates globally, with manufacturing plants located across the Americas, Asia, Europe, the Middle East, and Africa. DowDuPont’s commitment to innovation and sustainability has positioned it as a key player in the global market.
Business Overview
DowDuPont’s business is built on a foundation of scientific expertise and technological innovation. The company offers a wide range of products and solutions, from advanced materials to specialty products, catering to various sectors. In the healthcare industry, DowDuPont is particularly known for its high-quality medical device packaging solutions. These solutions are designed to protect medical devices from contamination and ensure their sterility, making them essential for healthcare providers and manufacturers.
Product Offered
DowDuPont’s medical device packaging products include Tyvek® 1073B and Tyvek® 1059B. Tyvek® 1073B is made from high-density polyethylene and offers superior protection against damage, making it ideal for high-risk medical products. Tyvek® 1059B, on the other hand, is designed for medium-risk applications and provides excellent microbial barrier properties. Both products are known for their durability, compatibility with various sterilization methods, and ability to maintain the sterility of medical devices.
Berry Plastics
Company Profile
Berry Plastics Corporation is a leading U.S. manufacturer of injection-molded plastic packaging, with a history dating back to 1967. The company has a strong presence in the Americas, Europe, Asia, and Africa, offering a wide range of plastic packaging solutions. Berry Plastics is known for its commitment to quality and innovation, providing solutions that meet the highest standards of safety and efficiency.
Business Overview
Berry Plastics operates in a variety of industries, including healthcare, food and beverage, and consumer goods. The company’s core product lines include aerosol over caps, open top containers, drink cups, custom molding, and housewares. In the healthcare sector, Berry Plastics specializes in medical device packaging, offering solutions that protect products, patients, and professionals. The company’s ISO 9001-certified facilities ensure that all products meet stringent quality standards.
Product Offered
Berry Plastics’ medical device packaging products include adhesive tapes, forming and pouch films, laminations, and nonwoven fabrics. These products are designed to provide robust protection for medical devices, ensuring they remain sterile and safe for use. Berry Plastics’ solutions are engineered to meet exacting specifications, making them ideal for a wide range of medical applications.
WestRock Company
Company Profile
WestRock Company is a leading provider of fiber-based paper and packaging solutions, established in 2015. The company operates through two main segments: Corrugated Packaging and Consumer Packaging. WestRock’s global presence includes manufacturing facilities in the Americas, Europe, and Asia, allowing it to serve customers worldwide. The company is known for its commitment to sustainability and innovation, providing solutions that meet the evolving needs of the market.
Business Overview
WestRock’s business is built on a foundation of fiber-based solutions, offering a wide range of packaging products and services. The company’s Corrugated Packaging segment includes containerboard mills, corrugated packaging, and distribution operations, while the Consumer Packaging segment focuses on innovative solutions for various industries. In the healthcare sector, WestRock provides specialized packaging for medical devices, ensuring they meet regulatory standards and maintain their integrity.
Product Offered
WestRock’s medical device packaging products include folding cartons, specialty labels, literature & booklets, adherence packaging, multiple component kits, and printed foils. These solutions are designed to provide value throughout the product life cycle, from manufacturing to end use. WestRock’s deep understanding of the healthcare industry allows it to stay ahead of regulatory changes and offer innovative packaging solutions that meet the highest standards of quality and safety.

